Revolutionary Reversible Anticoagulation
On-Demand Thrombin Control – The First Anticoagulant With a Built-In Molecular Off Switch
Revolutionary Reversible Anticoagulation
On-Demand Thrombin Control – The First Anticoagulant With a Built-In Molecular Off Switch
AYA1809002 is a first-in-class, reversible DNA aptamer-based anticoagulant targeting thrombin with high affinity and tunable control via a sequence-paired antidote. It is engineered to provide safe, effective, and rapidly reversible anticoagulation for use in hospital/acute care settings and high-risk patient populations.
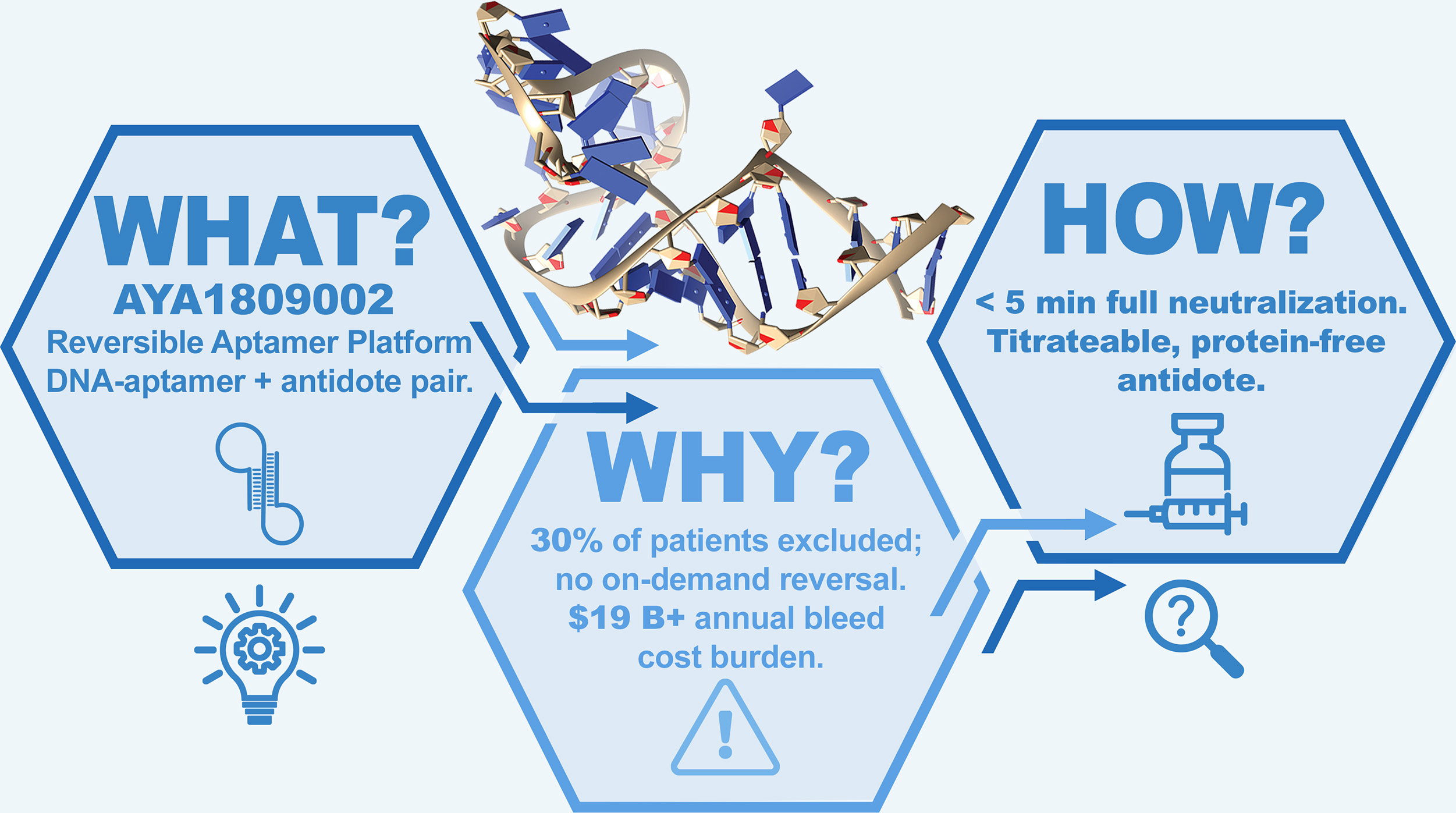
Mechanism, Selectivity & Pharmacokinetics
AYA1809002 is a highly selective thrombin inhibitor that binds Exosite I, the regulatory domain responsible for fibrin formation and platelet activation. By targeting this site rather than the catalytic active site, the aptamer selectively disrupts thrombin–substrate interactions while preserving catalytic structure.
This mechanism delivers potent anticoagulant activity with exceptional selectivity and predictable pharmacokinetics, supporting controlled and reliable anticoagulation.
Built-in Reversibility
AYA1809002 incorporates a paired DNA antidote system, enabling rapid and precise control of anticoagulation. A complementary oligonucleotide binds the aptamer in a 1:1 interaction, eliminating thrombin affinity and reversing anticoagulant activity within minutes. Because reversal is driven by molecular pairing, anticoagulation can be rapidly and precisely titrated to the desired level.
Direct Thrombin Inhibition
Comparison of Anticoagulant Approaches
Clinical and Economic Impact of Major Bleeding
Major bleeding remains a significant challenge in anticoagulated patients.
Intracranial hemorrhage occurs in ~0.5–1% of patients annually
30-day mortality can approach 50%
Major bleeding events can add $25,000–$60,000 in hospital costs and increase length of stay
Therapies that allow rapid and precise control of anticoagulation may help reduce these clinical and economic burdens.
AYA1809002 Development Timeline (Projected)

